In CRO and biotech marketing, keyword research is not a volume game. It is a precision game.
Most companies in this space operate in narrow markets, serve highly specialized buyers, and sell services that are expensive, technical, regulated, and trust-sensitive. That changes everything. A keyword with modest search demand can still be commercially exceptional if it signals sponsor intent, vendor evaluation, or a need tied to a specific clinical, regulatory, or laboratory milestone. Industry definitions and regulatory frameworks reinforce this reality: a CRO is formally understood as an organization performing trial-related duties on behalf of a sponsor, while the sponsor still retains ultimate responsibility for quality and oversight. That means buyers do not just search for “providers.” They search for proof, risk reduction, process quality, and operational credibility.
A serious keyword strategy for CRO and biotech companies therefore has to do more than generate topic ideas. It has to mirror the way sponsors, clinical operations teams, regulatory stakeholders, procurement teams, and scientific decision-makers actually think. It has to translate service lines, therapeutic areas, development stages, and compliance language into a search architecture that attracts the right visitors at the right point in the buying journey. Research on B2B buying behavior shows that modern buyers conduct substantial independent research before engaging sales, which makes search visibility and educational authority far more important than many life sciences firms assume.
Why keyword research works differently in CRO and biotech
The first mistake many marketers make is importing a generic SaaS or broad B2B SEO playbook into a life sciences environment. That does not hold up.
CRO and biotech search behavior is shaped by regulatory terminology, scientific specificity, internal buying committees, and long sales cycles. A sponsor does not always start with a brand name. Often, the search begins with a problem: how to structure an IND-enabling program, how to evaluate a Phase I unit, how to outsource bioanalytical validation, how to manage Phase II–III operations, or how to find regulatory submission support. Definitions from FDA, ClinicalTrials.gov, NCATS, and GLP/GCP authorities show how standardized this language already is across the industry.
That is why keyword research in this market must begin with industry taxonomy, not brainstorming. If your keyword universe is not anchored in actual service categories, trial phases, regulatory milestones, and scientific terminology, you will end up publishing content that ranks for curiosity instead of revenue.
Start with a service-led taxonomy
The strongest CRO and biotech keyword strategies are built from the inside out. Start with the real structure of the business.
For most CROs and biotech service providers, that means organizing keyword research around categories such as preclinical and nonclinical services, GLP toxicology, IND-enabling work, Phase I through Phase IV clinical operations, biometrics, data management, biostatistics, bioanalytical services, biomarkers, central lab capabilities, regulatory affairs, eCTD publishing, pharmacovigilance, medical writing, and therapeutic-area specialization. These are not just website sections. They are the foundation of how the market searches. FDA materials define IND and NDA processes, NCATS and ClinicalTrials.gov define clinical phase terminology, and EMA and OECD define GLP as a quality system for nonclinical safety studies.

From there, build keyword clusters around each service line. For example, a preclinical cluster may include terms around GLP toxicology studies, toxicokinetics, safety pharmacology, IND-enabling programs, and nonclinical study design. A clinical operations cluster may expand into Phase II oncology CRO, risk-based monitoring, site feasibility, decentralized trial operations, and patient recruitment strategy. A bioanalytical cluster may include biomarker assay development, ligand-binding assay validation, immunogenicity testing, and sample stability studies.
This is where mediocre SEO teams go wrong: they stop at the obvious head terms. Stronger teams keep going until they map the specific operational language that buyers use during planning and evaluation.
Use the language of science, not just the language of marketing
In biotech and CRO SEO, synonym control matters more than usual.
Scientific and regulatory topics often have multiple naming conventions, abbreviations, and related terms. That makes controlled vocabularies extremely valuable. MeSH, maintained by the U.S. National Library of Medicine, is especially useful for identifying related biomedical terminology, normalizing synonyms, and building clusters around concepts that may be searched in more than one way.
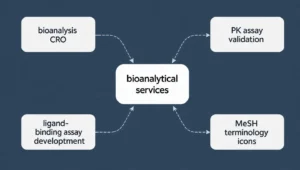
This matters because search demand in life sciences is often fragmented. One audience may search “bioanalytical lab services,” another “bioanalysis CRO,” another “PK assay validation,” and another “ligand binding assay development.” They may all belong in the same strategic cluster, but they will not appear that way unless you deliberately connect them.
That is also why search volume alone is a terrible decision-maker in this sector. Long-tail phrases with low apparent demand can still be high-intent, high-value opportunities, particularly when they align with outsourcing needs, program milestones, or vendor selection. Standard keyword research frameworks from Ahrefs, Moz, and Semrush all reinforce the importance of looking beyond head terms and understanding the relationship between keyword specificity, intent, and opportunity.
Map keywords to buying-stage intent
A professional keyword strategy does not treat all queries equally. It maps them to intent.
In this industry, the cleanest model is a B2B funnel that runs from discovery to consideration to evaluation to conversion.
At the discovery stage, users search for definitions, frameworks, requirements, and explanations. These are the “what is IND,” “clinical trial phases,” “what is GLP,” or “bioanalytical method validation explained” searches. They are educational, but they matter because they are often the first point of entry into a future commercial relationship. Search intent frameworks from Semrush and Ahrefs support this distinction between informational, commercial, navigational, and transactional query types.
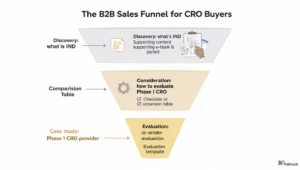
At the consideration stage, searches become more solution-aware. This is where you see phrases like “IND-enabling program checklist,” “risk-based monitoring approach,” “how to evaluate a Phase I CRO,” or “central lab setup for oncology trials.” These users are no longer just learning. They are designing a path forward.
At the evaluation stage, the language becomes vendor-driven and commercially serious. Queries include “Phase 1 CRO,” “bioanalytical lab services provider,” “regulatory affairs outsourcing,” “clinical data management CRO,” “Phase II oncology CRO,” or even “CRO proposal request.” These are the terms that deserve some of the highest priority in your content and landing-page strategy because they sit closest to opportunity creation.
The best keyword strategies do not force every term into a blog post. They map each cluster to the format the SERP and the buyer actually demand. Some keywords belong on service pages. Some deserve hub pages. Some need comparison content. Some belong in glossaries. Some should be supported by case studies or capability statements.
Build from first-party data before buying more tools
There is a blunt truth here: too many teams jump into third-party platforms before they have exhausted their own search data.
Google Search Console should be one of the first sources you use because it shows real query, click, impression, CTR, and average-position data for your site. That is not hypothetical demand. It is evidence of how search engines already interpret your content and where your current visibility is underperforming. Google documents these reporting metrics directly.
Google Keyword Planner is useful for rough demand validation, directional volume, and commercial clues such as CPC patterns, but it remains an advertising-oriented tool and should not be treated as the final authority on SEO opportunity. Google’s own documentation frames it in forecasting and planning terms.

Google Trends can help you detect relative shifts in interest, seasonality, or emerging modality-led themes, but Google also notes that Trends is based on sampled search data rather than absolute totals. That means it is useful for directional signal, not precise sizing.
Third-party platforms like Ahrefs and Semrush are still extremely useful, especially for competitor discovery, keyword-gap analysis, difficulty estimates, and SERP inspection. But they are best used after your internal query data and business taxonomy are already in place. Ahrefs and Semrush both provide strong workflows for competitor keyword analysis and keyword expansion, and those workflows are particularly valuable in niche B2B markets where you need to find topic gaps efficiently.
Run competitor analysis with discipline
Competitive keyword analysis is powerful, but lazy marketers misuse it constantly.
The goal is not to copy what competitors rank for. The goal is to identify where they have captured commercially relevant demand that you have not, then decide whether you can credibly compete.
That last part matters. In CRO and biotech, credibility is not decorative. It is decisive.
If a competitor ranks for a service term you do not cover, ask three questions. First, is the topic directly tied to your actual offer? Second, what type of page is winning in the SERP: a service page, a long-form guide, a glossary, or a comparison asset? Third, do you have the proof required to publish on that topic credibly, such as scientific depth, case evidence, process transparency, or compliance language? Keyword intent and SERP reality must drive the decision, not envy. Ahrefs’ competitor keyword workflows are useful here, but they still need human judgment.
This is particularly important for CROs that want to expand into adjacent topics. Ranking for a term your business cannot support operationally is not a growth tactic. It is a credibility leak.
Treat trust as part of the keyword strategy
In life sciences, trust is not a conversion optimization layer added later. It is part of the SEO strategy itself.
Google’s guidance on helpful content emphasizes people-first content, and Google’s Search Quality Evaluator Guidelines make clear that health-related and other YMYL topics demand especially high standards of experience, expertise, authoritativeness, and trust.
That has major implications for CRO and biotech content. A page targeting “IND-enabling services” cannot just repeat keywords and offer a generic sales pitch. It should demonstrate understanding of the regulatory pathway, define the program scope clearly, explain deliverables, describe quality and oversight processes, and support claims with transparent expertise signals. The same goes for pages targeting biomarker development, data management, clinical operations, or regulatory submissions.
In other words, the keyword tells you what the page is about. The trust architecture determines whether the page deserves to rank and whether a serious buyer will act on it.
That trust architecture may include expert bylines, citations to regulators and scientific bodies, capability statements, case studies, author bios, quality-system references, and plain-language explanations of process. In this market, that is not overkill. That is table stakes.
Design content around commercial depth, not just traffic potential
A strong CRO or biotech content strategy usually needs four content layers.
The first layer is service pages, which target the highest-value commercial and transactional terms. These are your money pages: “bioanalytical lab services,” “Phase II–III clinical operations,” “regulatory affairs outsourcing,” and similar phrases.
The second layer is topical hubs, which support service lines with broader yet still commercially aligned content. Examples include “IND-enabling programs,” “risk-based monitoring,” “oncology trial operations,” or “central lab strategy.”
The third layer is glossary and educational content. This captures discovery traffic and helps build topical authority around technical and regulatory terminology. Definitions of IND, NDA, GLP, clinical trial phases, biomarker-related concepts, and submission processes all belong here. FDA, NCATS, ClinicalTrials.gov, and other authoritative bodies provide reliable source foundations for this type of content.
The fourth layer is proof content: case studies, publications, posters, technical notes, and capability documents. These assets may not always be the first entry point from search, but they are often essential in moving a buyer from interest to trust.
That layered structure also supports internal linking. Google’s documentation makes clear that crawlable links and strong site structure help both users and search engines understand content relationships.
Account for international and regional search behavior
Many CRO and biotech firms work across markets, but their SEO strategies are often lazy about localization.
Translating pages is not the same as localizing search behavior. Different regions may use different terms for trial phases, services, outsourcing models, or regulatory pathways. If you operate internationally, your keyword research has to reflect actual local-language demand, not just English-language terminology copied into translated pages.
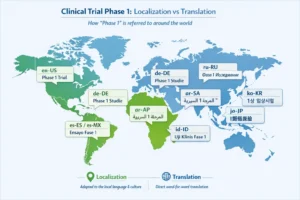
Google’s guidance on localized versions and hreflang supports a structured approach to language and regional targeting, including use of x-default where appropriate. Google also recommends clear URL structures for international content.
The practical implication is simple: English may remain the strategic core, but regional market pages need their own keyword validation and terminology standards. Otherwise, you end up with technically correct translations that no one actually searches.
Do not ignore technical SEO for scientific content
This sector tends to accumulate messy content assets: PDFs, duplicated regional pages, gated content, fragmented microsites, and nonstandard document libraries.
Technical SEO therefore matters more than many biotech marketers realize.
Google’s documentation on canonicalization, duplicate URL consolidation, sitemaps, robots instructions, and indexable file types is highly relevant for life sciences sites. Canonical signals help consolidate duplicate or near-duplicate pages. Sitemaps support discovery and can assist with multilingual implementations. Robots instructions and noindex rules need to be handled carefully, especially when important assets exist as PDFs or other non-HTML files. For non-HTML resources, Google recommends X-Robots-Tag rather than relying on meta robots tags.

This is not a small issue. Many CRO and biotech companies bury high-value capability information in PDFs, gate their strongest assets without providing indexable summaries, or let duplicate content spread across country folders and campaign microsites. That weakens visibility and creates avoidable crawl and indexing problems.
Structured data can also help reinforce content clarity. Google supports structured data types such as Article and Organization, which are relevant for many biotech and CRO sites publishing expert-led content.
Core Web Vitals and broader page experience are also worth monitoring, especially for large content hubs and lead-generation pages. Google explicitly uses Core Web Vitals within its ranking systems, even if they are not the sole driver of performance.
Measure success by pipeline relevance, not vanity metrics
The final test of a keyword strategy is not whether it produces more rankings. It is whether it produces more commercially relevant search visibility.
For CRO and biotech companies, that means measuring keyword success across several layers: visibility, traffic quality, conversion activity, and pipeline impact.
At the visibility layer, track impressions, clicks, CTR, and average position in Search Console.
At the traffic-quality layer, separate branded from non-branded growth and track how much organic traffic reaches core service pages, topic hubs, and evaluation-stage assets.
At the conversion layer, measure requests for proposals, consultation requests, demo or discovery calls, capability downloads, and other meaningful micro- and macro-conversions.
At the business layer, connect SEO to qualified pipeline wherever possible. In this sector, a keyword that generates a handful of serious opportunities may be far more valuable than a topic that drives thousands of irrelevant visits.
That is the point many teams miss. Keyword research is not about filling a content calendar. It is about aligning search demand with revenue logic.
The strategic takeaway
A winning keyword research strategy for CRO and biotech companies combines five disciplines.
It begins with a rigorous service taxonomy rooted in the real structure of clinical, regulatory, and laboratory work. It expands through scientific and regulatory language, not just generic marketing terms. It maps keywords to buyer intent and matching page formats. It treats trust, expertise, and compliance as part of SEO rather than separate concerns. And it measures success based on commercially meaningful outcomes rather than superficial traffic.
That is the level required in this market. Anything less is amateur SEO dressed up as life sciences strategy.
Need help turning this into pipeline, not just content?
If your CRO or biotech company needs an SEO and demand generation strategy that is built for complex B2B buying journeys, technical subject matter, and high-intent organic growth, Demand Enchance can help.
Demand Enchance supports brands that need more than blog production. The agency helps companies build serious search strategies around commercial intent, content architecture, SEO execution, conversion pathways, and market-specific messaging. For CRO and biotech firms, that means translating scientific expertise into discoverable, credible, revenue-oriented search visibility.
If your current keyword strategy is too broad, too generic, or too disconnected from sales reality, that is exactly the kind of problem Demand Enchance is built to fix.